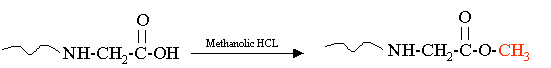
Making Methyl Esters
|
The methylation reaction adds a methyl group, +14 amu, to acidic groups on proteins. This reagent will react with aspartic acid, glutamic acid, and S-carboxymethylated cysteine. This reaction can be done to count the number of acidic residues in proteins and peptides. For example if a peptide had two aspartic acid residues the mass change after the reaction would be +42 amu. Two methyl esters would form on the acidic side chains of the two aspartic acids and one would form on the C-terminus of the peptide. Methylation can be used to confirm that the C-terminus of a peptide is intact and unmodified. Methylation is also used in MS/MS peptide sequencing to mark the acidic residues. A fragmentation is performed on the modified and unmodified peptide and the location of the acidic residue is obvious. In addition Glu only differs in mass from Gln and Lys by one mass unit, the addition of a methyl group removes any ambiguity. Beyond peptide sequencing, functional groups like carboxyl groups and primary amines exist on many small molecule drugs and we should keep in mind that we can explore the state of these groups through chemistry and mass spectrometry. |
|||||
| Back to Methylation | |||||
| - | |||||
| - | |||||
|
home | disclaimer Copyright © 3/1/2000 IonSource.Com All rights reserved. Last updated: Saturday, February 17, 2007 10:20:58 PM |
|||||