|
Mass
Spectrometry Standards
return
to standards index | return to
IonSource
Horse Myoglobin
Trypsin Digestion of Horse Apomyoglobin
Intro:
A trypsin digest of apomyoglobin is easy to make, and useful as a peptide
mapping system suitability standard. Below are a number of points why
myoglobin digest is our favorite peptide standard.
- A trypsin digest of myoglobin is inexpensive to prepare, and stable.
- A reduction and
alkylation is unnecessary, because horse myoglobin lacks cysteine, this is also true for
bovine beta casein. The
lack of cysteines makes the
digest protocol extremely simple for these two proteins.
- A myoglobin trypsin digest results in relatively few peptides. If
you run this standard daily, you soon become familiar with the peptides,
and their elution order. With such a simple mixture you
can tell at a glance if your mass spectrometer is performing properly.
- Relatively few large pieces are left after the
digest, making a greater
percentage of the peaks LC/MS and MS/MS friendly, this is not so true for
beta casein.
Note: Myoglobin containing the
heme is difficult to digest, apomyoglobin,
or myoglobin without the heme, is easy to digest. Myoglobin with the
heme intact is brown in color, because of the iron. You can acidify
the myoglobin to have it release the heme and then purify it away, but it is
easier to just buy apomyoglobin.
Materials:
Our vendor of choice for intact myoglobin is
Sigma-Aldrich
They sell 60 nmol vials of horse apomyoglobin,
Sigma-Aldrich product number A8673-1VL and A8673-5X1VL
. Sigma-Aldrich web
reference
Trypsin: Any trypsin will do, we routinely use
Promega modified
trypsin, but
Worthington bulk trypsin
works just as well. Trypsin autolysis
products will be an issue when you start doing protein ID, where the enzyme
to substrate ratio is typically higher.
Protocol:
- Reconstitute a 60 nmol vial of horse apomyoglobin in 6 ml of 10 mM
ammonium bicarbonate
(NH4HCO3).
The ammonium bicarbonate is not adjusted for pH.
Tris
base, pH 8, can be
substituted for ammonium bicarbonate in this step.
- Add 20ugs of Promega modified trypsin, and let sit on the
bench top, or in a drawer at ambient temperature. By the next morning,
15-18 hrs,
your digest will be ready to use. We have not found it necessary
to place the digest at 37C. Trypsin will act slower at ambient
temperature, but overnight is more than enough time for the reaction to
go to completion. In addition the elevated temperature, 37C, will
accelerate oxidations
carbamylations, and
deamidations,
Method Notes:
- Always use apomyoglobin, since the myoglobin with the intact heme
does not digest well.
- 60 nmols of myoglobin is about 1017 ug.
- The enzyme to substrate ratio in this procedure is approximately 1:50 (20ug/1017ug).
- The final concentration of this digest is 10 pmol/ul
- We store this digest at -20C, aliquoted, and it seems to be good forever (1yr+).
How it is used as a standard:
- Add 2ul of this standard to 1 ml of your HPLC solvent A (water, 0.1% formic
acid) to get an ESI LC/MS standard that is 20 fmol/ul
- It is useful as an LC/MS system suitability standard. We
typically run this standard at 50 to 100 fmols per injection. If we are doing
very sensitive work (silver stained gel protein ID) we will typically
run this standard at 5 to 10 fmol. We routinely bracket our
samples with this standard, so we can say, "Our instrument was
performing before and after your sample."
- It is useful as an internal standard. If you have a difficult,
client... we mean project, you can dope the sample with ~5 fmols of
the myoglobin standard. Make sure first that myoglobin will not interfere
with the analysis. That is, make sure your client is not looking for
myoglobin. You will then learn to relish the moment when you can say,
"Where is your protein? There's my protein standard at 5 fmols!"
- Since the resulting standard is at such a high concentration you can
often dilute 1:10 or 1:100 for infusion to tune ESI mass
spectrometers, without any desalting. We typically use the digest
done in ammonium bicarbonate for infusion. We have not tried the digest
made in Tris for this purpose; the salt might interefere.
- It is even useful as a MALDI standard after dilution, without desalting.
Example of Myoglobin ESI MS Peptide Map
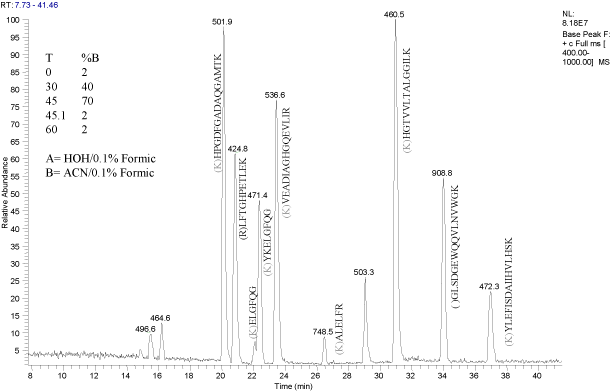
return
to standards index | return to
IonSource
|
home
| disclaimer
Copyright © 2000-2016 IonSource.Com All rights reserved.
Last updated:
Tuesday, January 19, 2016 02:49:53 PM
|

|